Welcome to the XuLab
The current research projects include:
- Design, Synthesis and Characterization of Innovative Bionanomaterials and Bionanophotonics.
- Development of Cutting-Edge Technologies and Instrumentation.
- Single Molecular and Single Live Cell Imaging.
- Single-Cell Genomics, Nanobiotechnology and Optogenetics, to unlock neuron-neuron communication, revolutionize early cancer detection, unravel developmental biology (embryonic stem cell differentiation), understand multidrug resistance, decipher nanotoxicity, design groundbreaking nanomedicines and biocompatible nanomaterials.
Tunable Rainbow Colored Nanoparticles (NPs)
 We design, synthesize and characterize a library of rainbow colored NPs with various sizes, shaped and surface properties, and study their physicochemical dependent optical properties and nanotoxicity at single NP resolution, aiming to rationally design biocompatible nanomaterials and optical probes for imaging cellular functions and for diagnosis and drug delivery. (J. Material Chemistry 20, 9867, 2010)
We design, synthesize and characterize a library of rainbow colored NPs with various sizes, shaped and surface properties, and study their physicochemical dependent optical properties and nanotoxicity at single NP resolution, aiming to rationally design biocompatible nanomaterials and optical probes for imaging cellular functions and for diagnosis and drug delivery. (J. Material Chemistry 20, 9867, 2010)
Single NP Microscopy and Spectroscopy
We have developed new microscopy and spectroscopy methods to image and characterize single NPs in real time. We have designed and developed single NP optical probes that resist photobleaching and blinking. We have used these photostable multicolored NPs as probes to detect single protein molecules (biomarkers) and study their functions in single live cells. Our goal is to develop a new class of photostable biocompatible NP Probes and imaging tools to study single live cells in real-time at the single-molecule and nm resolutions, aiming to depict their functions.
Single Molecule NP Optical Biosensors (SMNOBS) and Photostable Optical Nanoscopy (PHOTON)
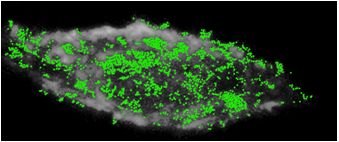 We design and synthesize SMNOBS to detect and characterize individual single protein molecules and their binding reactions in situ in real time. We develop PHOTON (new super-resolution optical nanoscopy) to image and track single protein molecules on/in single live cells and single embryos in real-time at the SM and nm resolutions. These new tools are enabling us to study the most significant and challenging questions in life sciences, aiming to understand how diseases develop, and to design effective approaches for disease prevention, diagnosis and treatments. (SMNOBS of a single TNFα molecule, each green dot, binds with its receptor on a single tumor cell, which leads to the cell death. We can determine how many TNFα molecules are required to initiate the death of a single tumor cell. Nanoscale, 12, 2797, 2012; JACS, 130, 17095, 2008; Anal. Chem. 79, 7708, 2007)
We design and synthesize SMNOBS to detect and characterize individual single protein molecules and their binding reactions in situ in real time. We develop PHOTON (new super-resolution optical nanoscopy) to image and track single protein molecules on/in single live cells and single embryos in real-time at the SM and nm resolutions. These new tools are enabling us to study the most significant and challenging questions in life sciences, aiming to understand how diseases develop, and to design effective approaches for disease prevention, diagnosis and treatments. (SMNOBS of a single TNFα molecule, each green dot, binds with its receptor on a single tumor cell, which leads to the cell death. We can determine how many TNFα molecules are required to initiate the death of a single tumor cell. Nanoscale, 12, 2797, 2012; JACS, 130, 17095, 2008; Anal. Chem. 79, 7708, 2007)
Mission Inside Single Live Cells
 We are using new tools (single NP probes, SMNOBS and PHOTON) that we have developed to image single live cells in real-time at the SM and nm resolutions, aiming to study functions of individual protein molecules in single live cells. For example, we are studying how membrane proteins can assemble efflux pumps (multidrug transporters) to extrude a wide range of drug molecules, leading to multidrug resistance (MDR). We are also studying how single ligand molecules can specifically bind with its receptor molecules on single cells to initiate signaling transduction pathways, which leads to cell death or uncontrollable cell growth (cancer). (Biochemistry 49, 5942, 2010; Anal. Bioanal. Chem. 397, 3317, 2010; Nano Letters, 2, 175, 2002).
We are using new tools (single NP probes, SMNOBS and PHOTON) that we have developed to image single live cells in real-time at the SM and nm resolutions, aiming to study functions of individual protein molecules in single live cells. For example, we are studying how membrane proteins can assemble efflux pumps (multidrug transporters) to extrude a wide range of drug molecules, leading to multidrug resistance (MDR). We are also studying how single ligand molecules can specifically bind with its receptor molecules on single cells to initiate signaling transduction pathways, which leads to cell death or uncontrollable cell growth (cancer). (Biochemistry 49, 5942, 2010; Anal. Bioanal. Chem. 397, 3317, 2010; Nano Letters, 2, 175, 2002).
Zebrafish Embryos as In Vivo Model Organisms and Assays
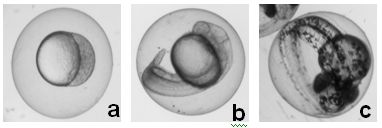 Zebrafish shares similar genetic phenotypes and drug binding sites as those in human. They are superior in vivo model organisms over others (mouse, rat, human). We are using our new imaging tools to study how zebrafish embryos develop, aiming to identify new biomarkers and pathways. We are also using zebrafish embryos as in vivo assays to screen biocompatibility and toxicity of nanomaterials, aiming to rationally design biocompatible nanomaterials and better understand effects of nanomaterials on living organisms. (Chem. Res. Toxico. 25, 1029, 2012; Nanoscale 1, 138, 2009; ACS Nano, 2, 1371, 2008; ACS Nano, 1, 133, 2007)
Zebrafish shares similar genetic phenotypes and drug binding sites as those in human. They are superior in vivo model organisms over others (mouse, rat, human). We are using our new imaging tools to study how zebrafish embryos develop, aiming to identify new biomarkers and pathways. We are also using zebrafish embryos as in vivo assays to screen biocompatibility and toxicity of nanomaterials, aiming to rationally design biocompatible nanomaterials and better understand effects of nanomaterials on living organisms. (Chem. Res. Toxico. 25, 1029, 2012; Nanoscale 1, 138, 2009; ACS Nano, 2, 1371, 2008; ACS Nano, 1, 133, 2007)
Study and Application of Nano/Micro-second Pulsed Electric Fields
Tuning membrane transport and subcellular structures of single live cells in real time holds great promise for selectively regulating cellular pathways, altering the fates of cells, and enabling smart drug delivery. We are developing new tools to study the effects of pulsed electric fields on the cellular functions, aiming to explore their new applications, including smart drug delivery and preparation of growth-arrested feeder cells for culture of embryonic stem cells (ESCs) (Biotechnol. J. 5, 588, 2010; J. Phys. Chem. B. 113, 14393, 2009).